GENOME EDITING AND ENGINEERING
Recent advancements in targeted gene editing and genome editing techniques have allowed scientists to choose from multiple molecular approaches for highly-specific, targeted DNA modifications to their gene(s) of interest in cell lines or mouse models. These techniques, including leveraging CRISPR/Cas9 nuclease, transcription activator-like effector-based nucleases (TALEN), and zinc finger nucleases (ZFNs), have allowed scientists to undertake genome editing experiments with unparalleled ease and specificity, ushering in a new era of widespread availability of genome engineering techniques and applications.
Cell Line Engineering and Genome Editing
Genome editing and gene editing techniques are increasingly being used in gene knockout and gene mutagenesis studies for engineering cell lines and mouse models. The CRISPR/Cas9 system in particular has been widely adopted to undertake gene knockout experiments in which a true knockout effect can be achieved in stable cell lines, as compared to traditional, transient and incomplete knockdown techniques such as RNAi. CRISPR technology is frequently leveraged for reversible gene knockdown, through CRISPR interference (CRISPRi) and gain-of-function gene knockin approaches are made possible by the CRISPR/Cas9 system as well. The precision and relative simplicity of the CRISPR gene editing system has enabled scientists to modify DNA quickly and efficiently in both engineered cell lines and mouse models, with a wide range of possible applications across research areas, target genomes, and industries. Gene editing has the potential to revolutionize genetic approaches to personalized medicine, and could enable a new era of gene-based therapeutic treatment options for heretofore untreatable diseases.
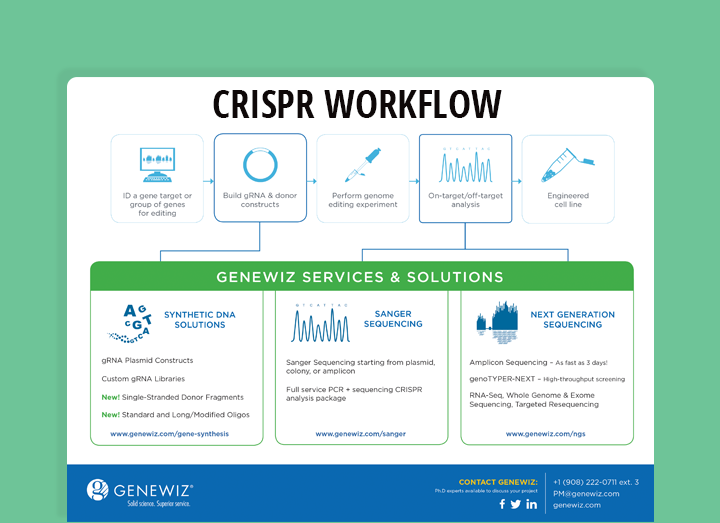
CRISPR Workflow and GENEWIZ Services
GENEWIZ is pleased to provide a number of services to expedite research in genome editing. The diagram below illustrates a typical CRISPR workflow and the steps within this workflow where GENEWIZ can help move your experiment forward.
Download NowGenome Editing in Research
Current examples of genome editing and gene editing applications in research include:
Cancer researchers investigate a candidate gene’s involvement in cancer progression using the CRISPR gene editing system to eliminate gene functionality and study the downstream effects in edited, stable cell lines.
Malaria and other mosquito-borne disease researchers leverage the CRISPR genome editing system to disrupt genes that control female mosquito reproduction, resulting in the possibility of a genetic approach to mosquito population control for regions susceptible to outbreaks of mosquito-borne diseases.
Stem cell researchers leverage CRISPR interference (CRISPRi) to reversibly knockdown expression of target genes in induced pluripotent stem cells (iPSCs) to study the effect on subsequent developmental pathways.
Biofuel crop scientists employ TALE-nucleases (TALENs) to disrupt highly redundant or polyploid lignin genes in the sugarcane genome to increase availability of cell wall sugars for enhanced production of ethanol in biofuel generation.